Hot injection thermolysis of heterometallic pivalate clusters for the synthesis of monodisperse zinc and nickel ferrite nanoparticles
Abstract
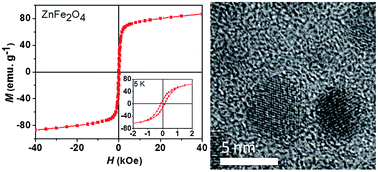
The heterometallic pivalate clusters- [Zn4Fe2O2(O2CtBu)10] (1) and [Fe2NiO(O2CtBu)6(HO2CtBu)3] (2) have been used as single source precursors to synthesise monodispersed zinc ferrite (ZnFe2O4) and nickel ferrite (NiFe2O4) nanoparticles respectively. The precursors were thermolysed with a mixture of oleylamine and oleic acid in either diphenyl ether or benzyl ether as the solvent at their respective boiling points of 260 and 300 °C. The effect of reaction time, temperature and concentration (0.25 or 0.50 mmol) on the stoichiometry, the phase or morphology of the nanoparticles were studied. TEM showed that highly monodispersed spherical nanoparticles of zinc ferrite (3.2 ± 0.2 nm) and nickel ferrite (3.3 ± 0.2 nm) respectively were obtained from (1) and (2) using 0.50 mmol precursor concentration at 260 °C. The decomposition of the precursors at 0.25 mmol and 300 °C gave larger nanoparticles of zinc ferrite (5.6 ± 0.5 nm) and nickel ferrite (5 ± 0.6 nm) from (1) and (2) respectively. The effect of reaction time was investigated for both precursors at 0.25 mmol by withdrawing aliquots at 5 minutes, 15 minutes, 30 minutes, 1 hour and 2 hours. Aliquots withdrawn at reaction times of less than 1 hour contain traces of iron oxide whilst only pure cubic zinc or nickel ferrite was obtained after one hour. Magnetic measurements revealed that all the ferrite particles are superparamagnetic at room temperature with high saturation magnetisation values. XMCD confirmed that in nickel ferrite particles, most of the Ni2+ cations are in the octahedral site. The hysteresis loop observed on the zinc ferrite nanoparticles indicated that there is cation redistribution, this is further evident in the XMCD analysis and EPMA result.
- This article is part of the themed collection: In memory of Paul O’Brien

 Please wait while we load your content...
Please wait while we load your content...