Azobenzene moiety variation directing self-assembly and photoresponsive behavior of azo-surfactants†
Abstract
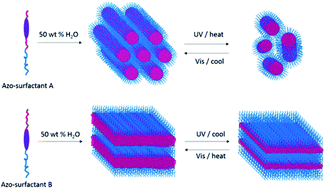
The effect of varying the position of the azobenzene group within two comparable photoresponsive amphiphiles on their capability to form lyotropic liquid crystals (LLCs) was investigated in detail in this study. Two photoresponsive amphiphiles having comparable structures were designed and synthesized consisting of hydrophilic oligooxyethylene units, a hydrophobic alkyl chain and a light-sensitive azobenzene moiety. When the azobenzene group was located in the middle of the hydrophobic alkyl chain, multiple LLC phases were observed at various water contents in the azo-surfactant–water binary system. In contrast, when the azobenzene group was directly attached to the hydrophilic domain, the azo-surfactant–water binary system exhibited only lamellar phases. The temperature dependence of these self-organised nanostructures was also investigated by the combination of small angle X-ray scattering (SAXS), differential scanning calorimetry (DSC), and rheology. Under alternating UV and visible light irradiation, reversible trans–cis photoisomerization of the azobenzene group occurred efficiently in dilute solution for both azo-surfactants. However, only photoisomerization of the surfactant possessing the azobenzene group localized in the middle of the alkyl chain induced significant changes in the self-assembled structure and its bulk properties. This study demonstrates that self-assembly and photoresponsive behaviour of photosensitive amphiphiles is extremely sensitive to the position of the photoactive moiety within the surfactant molecular architecture.

 Please wait while we load your content...
Please wait while we load your content...