Facile preparation, formation mechanism and microwave absorption properties of porous carbonyl iron flakes†
Abstract
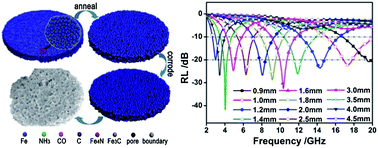
An easy and flexible two-step approach consisting of annealing and subsequently selective pitting corrosion has been developed to prepare porous carbonyl iron flakes (CIFs). Based on the dependence of the morphologies and compositions of the resultant CIFs on the annealing temperature and corrosion time, a formation mechanism for the porous CIFs has been substantiated. In our protocol, the annealing process carried out as the first step in the formation process causes an increase in the grain size and forces the Fe nanocrystals to react with the N impurities in the raw CIFs to generate dispersed Fe4N domains. These domains subsequently induce selective pitting corrosion, resulting in porous CIFs. As a result of the porous structure, the as-obtained porous CIFs have a specific surface area of 16.92 m2 g−1, a saturation magnetization of 140 emu g−1 and a coercivity of 61 Oe. Compared with the raw CIFs, they have a large permeability at high frequencies and a modest permittivity. Consequently, composites containing 20 vol% of the porous CIFs in paraffin wax display a reflection loss ≤−20 dB over a wide frequency range of 2.9–20 GHz when the thickness is varied between 0.9 and 4.5 mm. The results reported here suggest a facile approach to preparing porous nanocrystalline metals. Our results offer a promising strategy for the fabrication of absorbents for thin-thickness and strong-absorption microwave absorbing materials with working frequencies adjustable over a wide range simply by varying the thickness.

 Please wait while we load your content...
Please wait while we load your content...