Construction of efficient blue AIE emitters with triphenylamine and TPE moieties for non-doped OLEDs†
Abstract
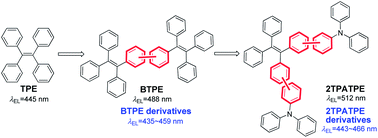
In this paper, by merging the hole-dominated triphenylamine (TPA) and tetraphenylethene (TPE) moieties together with different linkage positions, four derivatives of 1,2-bis[4′-(diphenylamino)biphenyl-4-yl]-1,2-diphenylethene (2TPATPE) were successfully synthesized with confirmed structures, and their thermal, optical and electronic properties were fully investigated. Thanks to the introduction of the meta-linkage mode on the TPE core, their π-conjugation length could be effectively restricted to ensure blue emission. The non-doped OLEDs based on these four emitters exhibit blue emissions from 443–466 nm, largely blue-shifted with respect to the green emission of 2TPATPE (514 nm). Meanwhile, good electroluminescence efficiencies with Lmax, ηC,max, and ηP,max of up to 8160 cd m−2, 3.79 cd A−1, and 2.94 Im W−1 respectively, have also been obtained, further validating our rational design of blue AIE fluorophores.

 Please wait while we load your content...
Please wait while we load your content...