A boron-dipyrrin–mercury(ii) complex as a fluorescence turn-on sensor for chloride and applications towards logic gates†
Abstract
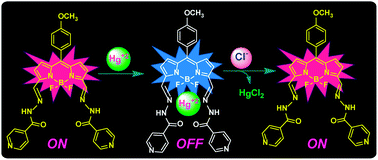
Chloride (Cl−) plays an important role in many cellular responses, including control of membrane potential, neurotransmission, regulation of cell volume and charge balance. Due to its physiological relevance, there is a growing need for improved optical sensors that can detect Cl− ions in biological and environmental samples. Herein, we have developed new N-acylhydrazone substituted BODIPY–mercury(II) based fluorescent turn-on sensors 1–Hg2+ and 2–Hg2+, which showed remarkable selectivity and specificity towards Cl− ions under physiological conditions. The new BODIPY compounds 1 and 2 were synthesized by treating their corresponding 3,5-diformyl BODIPYs 3 and 4 with isonicotinohydrazide in one step under simple reaction conditions. Compounds 1 and 2 absorb in 615–650 nm and emit in 625–660 nm regions. BODIPYs 1 and 2 showed exclusive sensing towards Hg2+ and formed 1–Hg2+ and 2–Hg2+ complexes, respectively, which resulted in the quenching of fluorescence. Job's plot analyses and HR-MS studies supported the formation of 1–Hg2+ and 2–Hg2+ complexes. Complexes 1–Hg2+ and 2–Hg2+ were found to be exclusive fluorescence turn-on sensors for chloride ions. Upon addition of Cl− ions to 1–Hg2+ and 2–Hg2+ complexes, the mercury(II) ions were extracted from the complexes by releasing the free BODIPYs 1 and 2 which reflected in the significant enhancement of fluorescence intensity (22-fold) with a detection limit of 108 nM. The reversibility and reusability of sensors for the detection of Hg2+ and Cl− ions were tested for six cycles. Interestingly, the sensor can be used to construct an IMPLIES logic gate system as demonstrated in this paper. Furthermore, the probe is cell membrane-permeable and can readily be used to detect the intracellular Cl− ions.

 Please wait while we load your content...
Please wait while we load your content...