Biodistribution and clearance of aminoclay nanoparticles: implication for in vivo applicability as a tailor-made drug delivery carrier†
Abstract
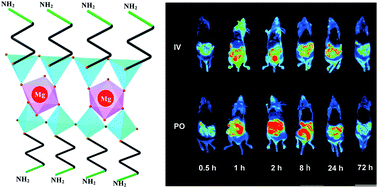
3-Aminopropyl functionalized magnesium phyllosilicate (aminoclay) is a tailor-made organoclay material that has many biomedical applications. Defining the biodistribution and in vivo pharmacokinetics of aminoclay is essential to gauge the therapeutic potential of aminoclay. Therefore, the present study investigated the in vivo fate of aminoclay nanoparticles in a mouse model. Fluorescent Cy5.0-conjugated-aminoclay (Cy5.0-aminoclay) was synthesized for optical imaging and quantification in vivo and then its tissue distribution as well as elimination pathways was investigated in mice. After intravenous administration, fast tissue distribution of Cy5.0-aminoclay into various organs including the liver, kidney, lung, heart, and intestine was evident, as was the rapid clearance from each organ without any long-term accumulation. In addition, systemically administered nanoparticles were eliminated mainly from the urine and feces, where the nearly complete recovery of administered dose was achieved within 72 h. After oral administration, the biodistribution and pharmacokinetic studies indicated no intestinal absorption of Cy5.0-aminoclay. Consequently, about 90% of orally administered nanoparticles were eliminated via feces within 24 h. Taken all together, the present study highlights the low risk for long-term tissue accumulation of aminoclay particles, which may be desirable attributes for biomedical applications as a drug delivery carrier.

 Please wait while we load your content...
Please wait while we load your content...