Double-layered hyaluronic acid/stearic acid-modified polyethyleneimine nanoparticles encapsulating (−)-gossypol: a nanocarrier for chiral anticancer drugs†
Abstract
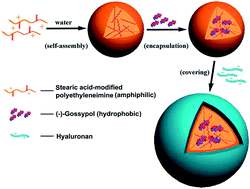
This study aimed to enhance the water solubility and antitumor efficacy of (−)-gossypol. Polyethyleneimine conjugated with stearic acid (PgS) was used for loading and protecting (−)-gossypol by hydrogen bonding. Double-layered hyaluronic acid (HA)-modified PgS nanoparticles encapsulating (−)-gossypol [(−)-G-PgSHAs] were prepared through a two-step fabrication process. The nanoparticles possessed a uniform spherical shape with a dynamic size of 110.9 ± 2.4 nm, which was determined through transmission electron microscopy and dynamic light scattering analysis. The encapsulation efficiency and drug loading capacity of (−)-G-PgSHAs were 72.6% ± 3.1% and 9.1% ± 0.42%, respectively. The IR spectra of the samples confirmed the protection effect of hydrogen bonding on the optical activity of the encapsulated (−)-gossypol. (−)-G-PgSHAs exhibited a controlled and tumor-specific release because of the high expression of HAase in the tumor region. The tumor-targeting feature of PgSHAs due to HA receptor mediation was confirmed by in vitro cell uptake and in vivo near infrared fluorescence imaging. The in vitro test showed that the (−)-G-PgSHAs had similar cytotoxicity to free (−)-gossypol and was lower than that of the encapsulated (±)-gossypol [(±)-G-PgSHAs]. The in vivo study of the anti-cancer effect of (−)-G-PgSHAs revealed that (−)-G-PgSHAs had a more enhanced tumor-suppression effect and reduced systemic toxicity compared with free (−)-gossypol and (±)-G-PgSHAs (P < 0.05). Therefore, PgSHA was a useful (−)-gossypol nanocarrier that exhibits high biocompatibility, tunable release of drug, and tumor-targeting characteristics for cancer treatment. In addition, this double-layered nanocarrier provided novel strategies for the encapsulation of other chiral drugs.

 Please wait while we load your content...
Please wait while we load your content...