Dually degradable click hydrogels for controlled degradation and protein release†
Abstract
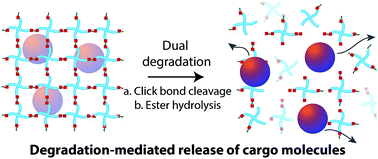
Click reactions have emerged as one of the most powerful paradigms in materials chemistry owing to their high regioselectivity and efficient reaction yields under mild conditions. While stability of the bonds formed by these reactions often is highly valued, their controlled cleavage is promising as an elegant approach to engineer material degradation for a number of applications, including drug delivery and tissue engineering. However, cleavage of click linkages under physiological conditions remains a major challenge in the design of degradable biomaterials. Here, we demonstrate the use of cleavable click linkages formed by Michael-type addition reactions in conjunction with hydrolytically cleavable functionalities for the degradation of injectable hydrogels by dual mechanisms for controlled protein release. Specifically, the reaction between maleimides and thiols was utilized for hydrogel formation, where thiol selection dictates the degradability of the resulting linkage under thiol-rich reducing conditions. Relevant microenvironments would include those rich in glutathione (GSH), a tripeptide that is found at elevated concentrations in carcinoma tissues. Degradation of the hydrogels was monitored with rheometry and volumetric swelling measurements. Arylthiol-based thioether succinimide linkages underwent degradation via click cleavage and thiol exchange reaction in the presence of GSH as well as ester hydrolysis, whereas alkylthiol-based thioether succinimide linkages only undergo ester hydrolysis. The resulting control over the degradation rate within a reducing microenvironment resulted in ~2.5 fold differences in the release profile of cargo molecules (fluorescently labeled bovine serum albumin as a model protein) from dually degradable hydrogels compared to non-degradable hydrogels. These unique degradable chemistries are promising for controlling the rate of degradation and local release of therapeutic cargo molecules in cancer tissue microenvironments.

 Please wait while we load your content...
Please wait while we load your content...