Backside-surface imprinting as a new strategy to generate specific plastic antibody materials
Abstract
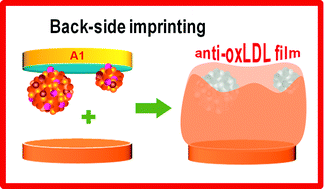
A backside protein-surface imprinting process is presented herein as a novel way to generate specific synthetic antibody materials. The template is covalently bonded to a carboxylated-PVC supporting film previously cast on gold, let to interact with charged monomers and surrounded next by another thick polymer. This polymer is then covalently attached to a transducing element and the backside of this structure (supporting film plus template) is removed as a regular “tape”. The new sensing layer is exposed after the full template removal, showing a high density of re-binding positions, as evidenced by SEM. To ensure that the templates have been efficiently removed, this re-binding layer was cleaned further with a proteolytic enzyme and solution washout. The final material was named MAPS, as in the back-side reading of SPAM, because it acts as a back-side imprinting of this recent approach. It was able to generate, for the first time, a specific response to a complex biomolecule from a synthetic material. Non-imprinted materials (NIMs) were also produced as blank and were used as a control of the imprinting process. All chemical modifications were followed by electrochemical techniques. This was done on a supporting film and transducing element of both MAPS and NIM. Only the MAPS-based device responded to oxLDL and the sensing layer was insensitive to other serum proteins, such as myoglobin and haemoglobin. Linear behaviour between log(C, μg mL−1) versus charged tranfer resistance (RCT, Ω) was observed by electrochemical impedance spectroscopy (EIS). Calibrations made in Fetal Calf Serum (FCS) were linear from 2.5 to 12.5 μg mL−1 (RCT = 946.12 × log C + 1590.7) with an R-squared of 0.9966. Overall, these were promising results towards the design of materials acting close to the natural antibodies and applied to practical use of clinical interest.

 Please wait while we load your content...
Please wait while we load your content...