Structure defects in g-C3N4 limit visible light driven hydrogen evolution and photovoltage†
Abstract
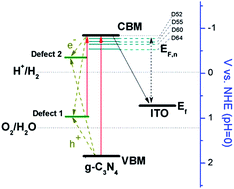
Graphitic carbon nitride (g-C3N4) is a promising visible-light-responsive photocatalyst for hydrogen generation from water. As we show here, the photocatalytic activity of g-C3N4 is limited by structure defects generated during the calcination process. Specifically we find that the photocatalytic hydrogen production rate from aqueous methanol is inversely related to the calcination temperature (520–640 °C). The highest activity of 0.301 mmol h−1 g−1 is observed for the sample prepared at the lowest processing temperature. Surface photovoltage (SPV) spectroscopy shows that the maximum photovoltage is reduced (from 1.29 V to 0.62 V) as the processing temperature is increased, in accordance with higher defect concentrations and faster electron–hole recombination. The defects also produce additional optical absorption in the visible spectra and cause a red shifted, weakened photoluminescence (PL). Based on the sub-gap signal in the SPV and PL spectra, defect energy levels are +0.97 V and −0.38 V (vs. NHE) in the band gap of the material. According to Fourier transform infrared (FTIR) spectra, the defects are due to amino/imino groups in the g-C3N4 lattice.


 Please wait while we load your content...
Please wait while we load your content...