Sodium deficient nickel–manganese oxides as intercalation electrodes in lithium ion batteries
Abstract
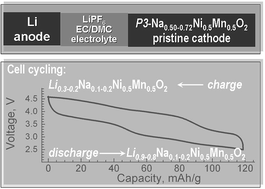
Sodium deficient nickel–manganese oxides NaxNi0.5Mn0.5O2 with a layered structure are of interest since they are capable of participating in reactions of intercalation of Li+ and exchange of Na+ with Li+. Taking into account the intercalation properties of these oxides, we provide new data on the direct use of NaxNi0.5Mn0.5O2 as low-cost electrode materials in lithium ion batteries instead of lithium analogues. Sodium deficient nickel–manganese oxides NaxNi0.5Mn0.5O2 are prepared at 700 °C from freeze-dried acetate precursors. The structure of NaxNi0.5Mn0.5O2 is analyzed by means of powder X-ray diffraction, SAED and HRTEM. The oxidation states of nickel and manganese ions are determined by X-ray photoelectron spectroscopy (XPS) and electron paramagnetic resonance spectroscopy (EPR). Model lithium cells are used to monitor the lithium intercalation into NaxNi0.5Mn0.5O2. The surface and composition stability of NaxNi0.5Mn0.5O2 during the electrochemical reaction is monitored by using ex situ XPS and LA-ICPMS. Layered oxides NaxNi0.5Mn0.5O2 exhibit a P3-type of structure, in which the solubility of sodium is limited between 0.5 and 0.75. At 700 °C, NaxNi0.5Mn0.5O2 consists of thin well-crystallized nanoparticles; some of the particles have sizes higher than 100 nm, displaying a trigonal superstructure. For all oxides, manganese ions occur in the oxidation state of +4, while the oxidation state of nickel ions is higher than +2 and depends on the sodium content. The electrochemical reaction occurs within two potential ranges at 3.1 and 3.8 V due to the redox manganese and nickel couples, respectively. During the first discharge, Li+ intercalation and Li+/Na+ exchange reactions take place, while the consecutive charge process includes mainly Li+ and Na+ deintercalation. As a result, all oxides manifest a reversible capacity of about 120–130 mA h g−1, corresponding to 0.5–0.6 moles of Li+. The formation of surface layers in the course of the electrochemical reaction is also discussed.

 Please wait while we load your content...
Please wait while we load your content...