Iron pyrite thin films deposited via non-vacuum direct coating of iron-salt/ethanol-based precursor solutions†
Abstract
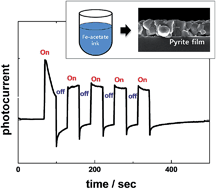
We report a new approach for fabricating pyrite (cubic FeS2) thin films via a non-vacuum direct solution coating route using iron-acetate dissolved in ethanol as a precursor solution. The precursor ink is deposited by spin coating and annealed in air at 300 °C to produce amorphous iron oxide films. Subsequent heat treatment of the iron oxide films in a sulfur environment at 450 °C results in the formation of phase-pure, carbon-free, large-grained pyrite films. In particular, the phase evolution during sulfurization is systematically investigated, focusing on the effects of the principal experimental variables such as temperature and pressure. As the temperature increases, iron oxide first begins to transform into marcasite (orthorhombic FeS2) and then is completely converted to pyrite. Further increasing the temperature above 500 °C induces the decomposition of pyrite into pyrrhotite (Fe1−xS) and sulfur vapor. The decomposition is also strongly dependent on the partial pressure of sulfur: as the pressure decreases, the decomposition occurs at lower temperature. The synthesized single-phase pyrite films show an indirect band gap of 0.94 eV, a strong anodic photocurrent based on photo-electrochemical measurements, and n-type semiconducting properties based on Mott–Schottky analysis. Thus, the films demonstrate great potential for use as absorbing layers in solar cells.

 Please wait while we load your content...
Please wait while we load your content...