First principles investigation of anion-controlled red shift in light absorption in ZnX (X = O, S, Se) nanocluster modified rutile TiO2
Abstract
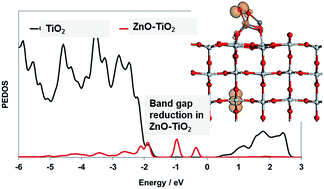
There is significant interest in finding new approaches to modifying TiO2 in order to endow it with visible light driven photocatalytic activity. To date, the most widely studied approach is to dope TiO2 with metals, non-metals or both simultaneously. However, there are some serious issues associated with doping of TiO2. We have studied an alternative approach in which TiO2 surfaces are modified with nanoclusters of metal oxides and have shown that this can introduce new states into the original TiO2 band gap that permits a red shift to induce visible light absorption while enhancing the photocatalytic activity over unmodified TiO2. In this paper we extend this idea to modification of rutile TiO2 with nanoclusters of ZnO, ZnS and ZnSe, which allows us to explore the possibility of tuning the magnitude of the red shift in modified TiO2 through the choice of anion in the nanocluster modifier. We find that the adsorption energies of the nanoclusters and the interfacial geometry are strongly influenced by the nature of the anion, with the ZnSe nanoclusters the most weakly bound, arising from the large ionic radius of Se giving longer surface–nanocluster distances and thus weaker binding. The magnitude of the red shift compared to unmodified rutile (110) can be over 1 eV and is tuned by the size (or surface loading) of the nanocluster and the anion in the nanocluster. Finally, a model of the photoexcited state shows that holes localised onto low coordinated anion sites on the adsorbed nanoclusters and electrons localise onto the TiO2 surface which will reduce charge recombination and enhance the photocatalytic activity. This concept of an anion controlled red shift in light absorption in nanocluster-modified TiO2 should be extendable to other metals such as tin or lead chalcogenides.

 Please wait while we load your content...
Please wait while we load your content...