Hierarchically porous materials built of Fe–silicalite nanobeads†
Abstract
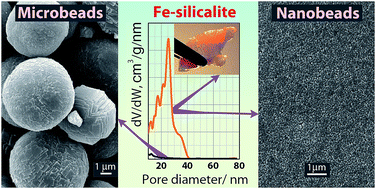
Hierarchically porous zeolite materials built of closely and randomly packed uniform Fe–silicalite nanobeads were synthesized. The desired assembly of nanobeads was achieved by the centrifugation and sedimentation of nanozeolite suspension followed by drying and calcination. A micro/meso/macroporous Fe–silicalite material with spongy texture built of closely packed nanocrystals was designed using polystyrene latex as a supramolecular template. Large Fe–silicalite microbeads were synthesized to compare their structure with the nanocrystalline materials. The synthesized samples were characterized by laser diffraction analysis, X-ray diffraction, scanning and transmission electron microscopy, energy-dispersive X-ray spectroscopy, argon and nitrogen adsorption measurements, inductively coupled plasma optical emission spectrometry, UV visible diffuse reflectance spectroscopy and temperature-programmed desorption of ammonia. Fe–silicalite nanozeolite materials have shown high crystallinity, and possess micro- and meso/macropore surface areas and high specific pore volumes. Pellets built of closely packed nanocrystals have exhibited good mechanical stability in benzene and water. Calcined Fe–silicalite materials built of nanobeads were observed to contain highly dispersed ferric clusters no more than 1 nm in size. The 3 nm ferric clusters in the zeolitic microbeads resulted in the appearance of Lewis acid sites with medium strength, which are absent in the nanobeads. Catalytic performance of hierarchically porous Fe–silicalites was studied in the total oxidation of clarithromycin lactobionate by H2O2 at 323 K compared with Fe–silicalite microbeads. Hierarchical Fe–silicalites are more efficient catalysts vs. Fe–silicalite microbeads due to increased catalytic site accessibility.
- This article is part of the themed collection: Porous Materials (FEZA 2014)

 Please wait while we load your content...
Please wait while we load your content...