Thermodynamic and kinetic assessments of strontium-doped lanthanum manganite perovskites for two-step thermochemical water splitting
Abstract
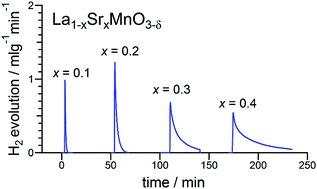
Solar-driven thermochemical water splitting using non-stoichiometric oxides has emerged as an attractive technology for solar fuel production. The most widely considered oxide for this purpose is ceria, but the extreme temperatures required to achieve suitable levels of reduction introduce challenges in reactor design and operation, leading to efficiency penalties. Here, we provide a quantitative assessment of the thermodynamic and kinetic properties of La1−xSrxMnO3−δ perovskites, targeted for a reduced temperature operation of thermochemical water splitting. Sr-doping into lanthanum manganite increases the thermodynamic fuel production capacity, which reaches 9 ml g−1 for 0.4 Sr for a thermochemical cycle operated between 1400 and 800 °C. The hydrogen yields are moreover in good agreement with expected values based on analysis and extrapolation of thermogravimetric data available in the literature. High levels of Sr doping, however, result in low steam-to-hydrogen conversion rates, implying high energy penalties in an operational reactor. Furthermore, the rate of fuel production decreases with increasing Sr content, suggesting that intermediate compositions may yield the most suitable combination of properties.

 Please wait while we load your content...
Please wait while we load your content...