Selective adsorption of contaminants on Pb(Zr,Ti)O3 surfaces shown by X-ray photoelectron spectroscopy
Abstract
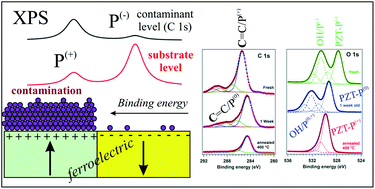
X-ray photoelectron spectroscopy (XPS) analyses of lead zirco-titanate Pb(Zr,Ti)O3(001) single crystal thin layers as a function of the time spent between sample preparation by pulsed laser deposition and introduction in to a ultrahigh vacuum revealed the fact that freshly prepared samples showed a shift of the C 1s towards a higher binding energy, together with shifts of core levels originating from the substrate (particularly Ti 2p and O 1s) towards a lower binding energy. This behaviour is explained by considering that the molecules of contaminants (fatty acids, alcohols, esters) are adsorbed preferentially on areas exhibiting outwards polarization P(+). Thus, photoelectrons originating from contaminants will have larger binding energies because of the charge state of the P(+) areas, whereas the substrate XPS signals from these P(+) areas are attenuated by the contaminants, with the consequence of a prevalence of XPS substrate signals originating from the P(−) areas, shifted towards lower binding energies. Piezoresponse force microscopy confirmed the assumptions derived from XPS results and suggests the existence of an interplay between the adsorption of contaminants and the surface polarization state.

 Please wait while we load your content...
Please wait while we load your content...