Tailoring hierarchically structured SiO2 spheres for high pressure CO2 adsorption†
Abstract
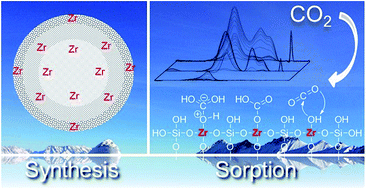
The synthesis, characterization, and experimental validation of hierarchically structured, millimeter-sized SiO2 spheres with high CO2 uptake capacities have been systematically explored. The solvents employed during synthesis (aniline, benzyl alcohol and butanol) of the silica structures from alkoxisilanes strongly influence the physicochemical properties by controlling hydrolysis and condensation rates. The new sorbents possess specific surface areas up to 660 m2 g−1 and a hierarchically ordered mesoporous/macroporous pore structure. The SiO2 spheres showed lower heat of adsorption of CO2 (8–17 kJ mol−1) compared to the benchmark zeolite 13X, facilitating the desorption of CO2 in temperature swing adsorption applications. Moreover, the CO2 adsorption isotherms of SiO2 spheres are less steep compared to those of zeolite 13X resulting in an increased CO2 uptake capacity in pressure swing adsorption processes. Addition of Zr4+ cations to SiO2 increases the CO2 uptake by generating Lewis acid–base sites. Because the presence of Zr4+ in the structure also dramatically enhances the abrasion resistance of the sorbents, additional coagulation steps, required for the benchmark sorbents, are not necessary.
- This article is part of the themed collection: Porous Materials (FEZA 2014)

 Please wait while we load your content...
Please wait while we load your content...