Exploiting stable radical states for multifunctional properties in triarylamine-based porous organic polymers†
Abstract
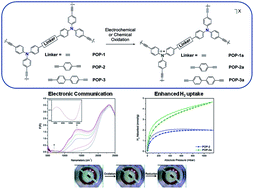
Redox-active porous organic polymers (POPs) have enormous potential in applications ranging from electrocatalysis to solar energy conversion. Exploiting the different electronic states offers exciting prospects for controlling host–guest chemistry, however, this aspect of multifunctionality has to date, remained largely unexplored. Here, we present a strategy for the development of multifunctional materials with industrially sought-after properties. A series of hydrophobic POPs containing redox-active triarylamines linked by ethynyl (POP-1), 1,4-diethynylphenyl (POP-2) and 4,4′-diethynylbiphenyl (POP-3) bridges have been synthesised and characterised by NMR and EPR spectroscopy, as well as spectroelectrochemistry and computational modelling. The facile electrochemical or chemical oxidation of the POPs generate mixed-valence radical cation states with markedly enhanced adsorption properties relative to their neutral analogues, including a 3-fold improvement in the H2 uptake at 77 K and 1 bar, and an increase in the isosteric heat of adsorption for CO2.

 Please wait while we load your content...
Please wait while we load your content...