Reduction of the oxygen reduction reaction overpotential of nitrogen-doped graphene by designing it to a microspherical hollow shape†
Abstract
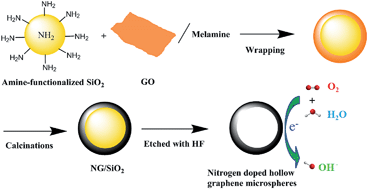
Nitrogen-doped hollow graphene microspheres (NHGSs), synthesized through a procedure involving the calcination of graphene–oxide-wrapped amine-functionalized mesoporous silica nanoparticles (AFMSNs) and the subsequent removal of the AFMSNs via HF etching, have been employed as an electrocatalyst for the oxygen reduction reaction (ORR). It has been shown that these NHGSs are highly active toward ORR and exhibit higher electrocatalytic activity than graphene, nitrogen-doped graphene (NG), hollow graphene microspheres (HGSs), and JM Pt/C 40 wt%, and a comparable overpotential to JM Pt/C 40 wt%. Their high electrocatalytic activity could be attributed to the N-doped graphitic structure, which produces more active sites for the ORR, allowing the easier adsorption of oxygen and the subsequent reduction; and the specific microspherical hollow structure, which promotes the exposure of more surface area accessible to electrolytes, thereby allowing an easier diffusion of electrolytes into and out of the electrode catalyst layers. Their low overpotential for the ORR can be attributed to the specific microspherical hollow structure, which reduces the overpotential contribution from the mass transport limitation. The stability measurements show that NHGSs also exhibit a much higher stability than graphene, NG, HGSs, and JM Pt/C 40 wt%, and are immune to the methanol crossover and CO poisoning effects, which all together makes NHGSs highly attractive as an electrocatalyst for the ORR.

 Please wait while we load your content...
Please wait while we load your content...