Enhanced thermoelectric performance of PEDOT with different counter-ions optimized by chemical reduction†
Abstract
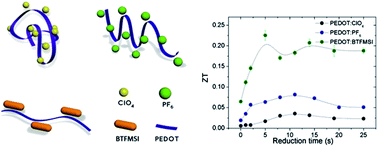
This work reports on the synthesis of the intrinsically conducting polymer poly(3,4-ethylenedioxythiophene) (PEDOT) doped with several counter-ions, ClO4, PF6 and bis(trifluoromethylsulfonyl)imide (BTFMSI), by electro-polymerization and its thermoelectric properties. We show that, depending on the counter-ion size, the thermoelectric efficiency of PEDOT can be increased up to two orders of magnitude. A further chemical reduction with hydrazine optimizes the power factor (PF). By changing the counter-ions, we were able to increase the electrical conductivity (σ) of PEDOT by a factor of three, while the Seebeck coefficient remains at the same order of magnitude in the three polymers. The best thermoelectric efficiency has been observed in PEDOT:BTFMSI. From the measurement of the Seebeck coefficient and σ, a PF of 147 μW m−1 K−2 has been deduced, while the measured thermal conductivity is κ = 0.19 W m−1 K−1, resulting in a ZT ∼ 0.22 at room temperature, one of the highest values reported in the literature for polymers. The increase in σ with the change of the counter-ion is mainly due to the stretching of the polymer chains. In this work, we provide a chemical route to further improve ZT in polymers and demonstrate a method of synthesis based on the electro-polymerization on gold. After removing the gold layer, a very thin semiconducting polymer film can be isolated.

 Please wait while we load your content...
Please wait while we load your content...