Nanoporous covalent organic polymers incorporating Tröger's base functionalities for enhanced CO2 capture†
Abstract
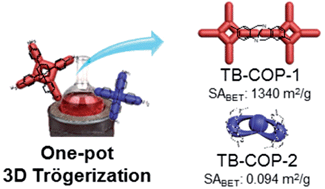
The CO2 uptake capacity and CO2/N2 selectivity of Tröger's base-bridged nanoporous covalent organic polymers (TB-COPs) were investigated. The TB-COPs were synthesized by reacting the terminal amines of tetrahedral monomers – namely, tetraanilyladamantane and tetraanilylmethane – with dimethoxymethane in a one-pot reaction under relatively mild conditions. Interestingly, these two tetrahedral monomers formed nanoporous polymers with substantially different surface areas. While the polymer resulting from the Trögerization of the tetraanilyladamantane monomer (TB-COP-1) exhibited a high surface area of 1340 m2 g−1, that from the tetraanilylmethane monomer (TB-COP-2) was found to be only 0.094 m2 g−1. This unusual phenomenon can be explained by the proximity of the amino moieties to each other within the monomeric unit. A shorter distance between the amino groups enables intramolecular as well as intermolecular cyclization, thus resulting in a much lower porosity. TB-COP-1 exhibited significant CO2 uptake capacities of up to 5.19 and 3.16 mmol g−1 at 273 and 298 K under ambient pressure, and CO2/N2 selectivities of 79.2 and 68.9 at 273 and 298 K at 1 bar for a gas mixture of CO2 : N2 at a ratio of 0.15 : 0.85. It is noteworthy that TB-COP-1 showed remarkable selectivity retention when increasing the temperature from 273 to 298 K.

 Please wait while we load your content...
Please wait while we load your content...