Rapid phosphine-free synthesis of CdSe quantum dots: promoting the generation of Se precursors using a radical initiator†
Abstract
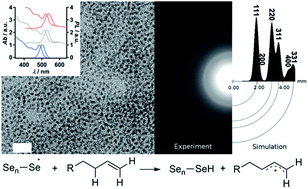
The replacement of phosphine containing compounds in the synthesis of II–VI quantum dots (QDs) via the ‘hot-injection’ method has received considerable attention in recent years, in particular toward scaling-up production. A key bottleneck in current approaches is the poor solubility of elemental Se in solvents such as 1-octadecene (1-ODE) or oleylamine requiring a heating temperature of 200 °C for several hours, or the introduction of additives such as alkylthiols or NaBH4, or replacement by SeO2. In the present work, we elucidate the fundamental steps in the dissolution of elemental Se in 1-ODE and oleylamine with the view of facilitating the large-scale synthesis of CdSe QDs. The main organoselenium species generated during the solubilisation of elemental Se in 1-ODE and oleylamine were identified by 1D and 2D NMR spectroscopy (1H, 13C and 77Se). Experimental evidence suggests that the rate determining step is the formation of Se radicals, via homolytic cleavage of the Se–Se bond, that attack the allylic proton in 1-ODE and oleylamine. Plausible reaction pathways in both systems are proposed. Finally, we demonstrate that the radical-mediated solubilisation of Se can be significantly accelerated by the addition of azobisisobutyronitrile (AIBN), a common radical initiator used in the polymer industry. In this way, a highly concentrated Se precursor was prepared. The “hot-injection” of the Se precursor into CdO containing 1-ODE solution leads to the formation of highly luminescent CdSe QDs with a well-defined cubic structure.

 Please wait while we load your content...
Please wait while we load your content...