Ultrahigh iodine adsorption in porous organic frameworks†
Abstract
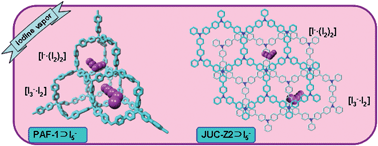
We present two porous organic frameworks (POFs), PAF-1 and JUC-Z2, with ultrahigh iodine capture capacity. The iodine vapor uptake of PAF-1 and JUC-Z2 were 1.86 g g−1 and 1.44 g g−1 respectively at 298 K per 40 Pa, which is extremely high for such low pressure sorption conditions. In addition, PAF-1 and JUC-Z2 could adsorb iodine over water with the selectivity of 5.1 and 6.5 respectively. The isosteric enthalpy at zero surface coverage, calculated by a virial equation with the iodine vapor sorption isotherms at 298 K and 313 K of JUC-Z2, reached −51.1 kJ mol−1, which was much higher than the coverage of PAF-1 (−14.9 kJ mol−1). Raman measurement confirmed the polyiodide to be I5− in POFs. Furthermore, solvents with different polarities, such as n-hexane, chloroform, and methanol, were chosen to conduct iodine binding measurements on PAF-1 and JUC-Z2. The formation constant Kf for POFs in n-hexane, chloroform and methanol drastically decreased with the increase in polarity, thus illustrating the important role of solvents in iodine binding.

 Please wait while we load your content...
Please wait while we load your content...