One-pot cation exchange synthesis of 1D porous CdS/ZnO heterostructures for visible-light-driven H2 evolution†
Abstract
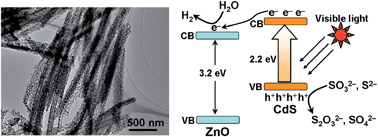
To investigate the size- and morphology-dependent properties of nanomaterials and to bring many of their potential applications into reality, inorganic nanomaterials with unique composition, morphology, size, and even heterojunction structures are still needed. Herein we report the synthesis of photocatalytically active, one-dimensional (1D), porous CdS/ZnO heterostructured nanomaterials with tunable aspect ratios. Moreover, we show that the as-obtained nanomaterials exhibit significantly enhanced visible light-activated photocatalytic activity compared to pure CdS toward hydrogen evolution reaction (HER). The synthesis of the materials is achieved via a facile one-pot cation exchange reaction (CER) between novel 1D ZnS/CHA (CHA = cyclohexylamine) inorganic–organic hybrid nanomaterials and Cd2+ ions. The two key steps for the synthesis being successful are: (1) the preparation of 1D ZnS/CHA nanomaterials with tunable aspect ratios and (2) the ability of the resulting nanomaterials to undergo CER (in Cd2+ solution) without completely leaving behind their exchangeable ions (i.e., Zn2+ in our case) in the solution. Consequently, some of the exchanged ions during CER are transformed into metal oxide (i.e., ZnO) nanoparticles in situ. This process ultimately leads to the formation of novel 1D CdS/ZnO heterostructured nanocomposite materials. Noteworthy also is that the as-obtained CdS/ZnO nanomaterials efficiently photocatalyze HER under visible light, affording an activity about 15 times higher than that of a porous pure CdS nanomaterial. The efficient photocatalytic activity of the former was attributed to their overall conducive structural features for HER, including their heterostructures at nanoscale.

 Please wait while we load your content...
Please wait while we load your content...