Real-time materials evolution visualized within intact cycling alkaline batteries
Abstract
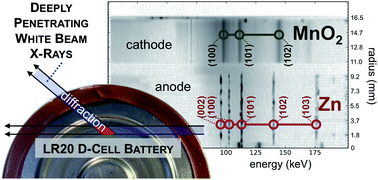
The scientific community has focused on the problem of inexpensive, safe, and sustainable large-scale electrical energy storage, which is needed for a number of emerging societal reasons such as stabilizing intermittent renewables-based generation like solar and wind power. The materials used for large-scale storage will need to be low cost, earth-abundant, and safe at the desired scale. The Zn–MnO2 “alkaline” battery chemistry is associated with one-time use, despite being rechargeable. This is due to material irreversibilities that can be triggered in either the anode or cathode. However, as Zn and MnO2 have high energy density and low cost, they are economically attractive even at limited depth of discharge. As received, a standard bobbin-type alkaline cell costs roughly $20 per kW h. The U.S. Department of Energy ARPA-E $100 per kW h cost target for grid storage is thus close to the cost of alkaline consumer primary cells if re-engineered and/or cycled at 5–20% nominal capacity. Herein we use a deeply-penetrating in situ technique to observe ZnO precipitation near the separator in an alkaline cell anode cycled at 5% DOD, which is consistent with cell failures observed at high cycle life. Alkaline cells designed to avoid such causes of cell failure could serve as a low-cost baseload for large-scale storage.

 Please wait while we load your content...
Please wait while we load your content...