A noble metal-free reduced graphene oxide–CdS nanorod composite for the enhanced visible-light photocatalytic reduction of CO2 to solar fuel
Abstract
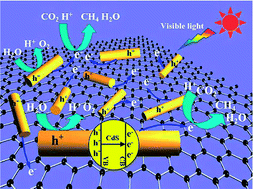
Solar-fuel production has attracted considerable attention because of the current demand to find alternative transportation fuels with particular emphasis on those fuels obtained photocatalytically from water and CO2. In this work, reduced graphene oxide (RGO)–CdS nanorod composites were successfully prepared by a one-step microwave-hydrothermal method in an ethanolamine–water solution. These composite samples exhibited a high activity for the photocatalytic reduction of CO2 to CH4, even without a noble metal Pt co-catalyst. The optimized RGO–CdS nanorod composite photocatalyst exhibited a high CH4-production rate of 2.51 μmol h−1 g−1 at an RGO content of 0.5 wt%. This rate exceeded that observed for the pure CdS nanorods by more than 10 times and was better than that observed for an optimized Pt–CdS nanorod composite photocatalyst under the same reaction conditions. This high photocatalytic activity was ascribed to the deposition of CdS nanorods onto the RGO sheets, which act as an electron acceptor and transporter, thus efficiently separating the photogenerated charge carriers. Furthermore, the introduction of RGO can enhance the adsorption and activation of CO2 molecules, which speeds up the photocatalytic reduction of CO2 to CH4. The proposed mechanism for the observed photocatalytic reaction with the RGO–CdS nanorod composite was further confirmed using transient photocurrent response and electrochemical impedance spectra. This work not only demonstrates a facile microwave-assisted hydrothermal method for fabricating highly active RGO–CdS nanorod composite photocatalysts, but also demonstrates the possibility of utilizing of an inexpensive carbon material as a substitute for noble metals in the photocatalytic reduction of CO2.
- This article is part of the themed collection: JMC A Top Picks collection: Harnessing the power of the sun

 Please wait while we load your content...
Please wait while we load your content...