An architectural development for energy conversion materials: morphology-conserved transformation synthesis of manganese oxides and their application in lithium ion batteries
Abstract
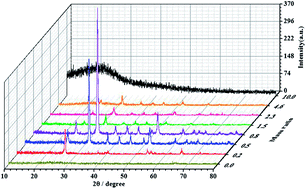
A facile and effective redox reaction is developed to build various morphologies and crystal phases of manganese-based compounds by using two kinds of raw material, D-maltose and KMnO4. Interestingly, not only different morphologies, but also different crystal phases could be precisely tailored by adjusting the mass ratio of D-maltose to KMnO4. After thorough analysis of the observations, a reasonable formation mechanism is proposed to offer physico-chemical insight, while promoting opportunities to explore novel properties of manganese oxides for the fabrication of important functional devices. To demonstrate the process–structure–property relationship of the as-prepared nanomaterials, various morphologies of α-Mn2O3, including cubic, spindle and fusiform were used for lithium ion batteries. The results indicate that the improved material morphology and porous structure can significantly improve the discharge capacity and cycling stability.

 Please wait while we load your content...
Please wait while we load your content...