Adsorption of carboxylic modified latex particles at liquid interfaces studied by the gel trapping technique
Abstract
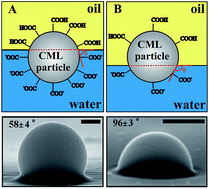
We have studied how carboxylic modified latex (CML) microparticles adsorb at liquid surfaces and the preferred type of emulsion they can stabilise depending on the particle size and the surface density of carboxylic groups. We measured the particle contact angle by using the gel trapping technique (GTT) for CML particles adsorbed at air–water and oil–water interfaces. Using this method we obtained scanning electron microscopy (SEM) micrographs of polydimethylsiloxane (PDMS) replicas of the liquid interface with the particles, where the PDMS replicates the non-polar phase and measured the particle contact angle. We discovered that the particle wettability correlates well with the surface density of the carboxylic groups but is not very sensitive to the presence of electrolyte in the aqueous phase and the value of the particle zeta potential. We demonstrated that CML microparticles with a high surface density of COOH groups stabilise oil-in-water (O/W) emulsions while those with the lowest coverage of COOH groups favour the formation of water-in-oil (W/O) emulsions. We found that this corresponds to a change of the CML particle contact angle from lower than 90° to higher than 90° upon decrease of the surface density of COOH groups. The findings confirm that the surface density of polar groups has a much bigger effect on the particle wettability and the preferred emulsion than the particle surface charge and zeta potential. Our results on the type of stabilised Pickering emulsion agree with other experimental studies with different particle materials. We propose an alternative explanation for the link between the particle contact angle and the type of stabilised Pickering emulsion.

 Please wait while we load your content...
Please wait while we load your content...