Formation and rupture of Ca2+ induced pectin biopolymer gels†
Abstract
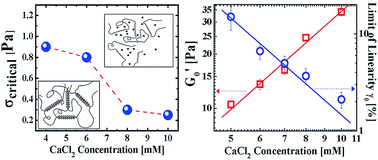
When calcium salts are added to an aqueous solution of polysaccharide pectin, ionic cross-links form between pectin chains, giving rise to a gel network in dilute solution. In this work, dynamic light scattering (DLS) is employed to study the microscopic dynamics of the fractal aggregates (flocs) that constitute the gels, while rheological measurements are carried out to study the process of gel rupture. As the calcium salt concentration is increased, DLS experiments reveal that the polydispersity of the flocs increase simultaneously with the characteristic relaxation times of the gel network. Above a critical salt concentration, the flocs become interlinked to form a reaction-limited fractal gel network. Rheological studies demonstrate that the limits of the linear rheological response and the critical stresses required to rupture these networks both decrease with the increase in salt concentration. These features indicate that the ion-mediated pectin gels studied here lie in a ‘strong link’ regime that is characterised by inter-floc links that are stronger than intra-floc links. A scaling analysis of the experimental data presented here demonstrates that the elasticities of the individual fractal flocs exhibit power-law dependences on the added salt concentration. We conclude that when both pectin and salt concentrations are increased, the number of fractal flocs of pectin increases simultaneously with the density of crosslinks, giving rise to very large values of the bulk elastic modulus.

 Please wait while we load your content...
Please wait while we load your content...