Chemoenvironmental modulators of fluidity in the suspended biological cell†
Abstract
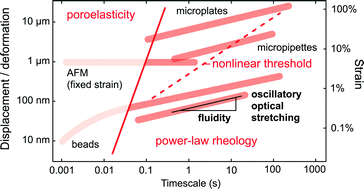
Biological cells can be characterized as “soft matter” with mechanical characteristics potentially modulated by external cues such as pharmaceutical dosage or fever temperature. Further, quantifying the effects of chemical and physical stimuli on a cell's mechanical response informs models of living cells as complex materials. Here, we investigate the mechanical behavior of single biological cells in terms of fluidity, or mechanical hysteresivity normalized to the extremes of an elastic solid or a viscous liquid. This parameter, which complements stiffness when describing whole-cell viscoelastic response, can be determined for a suspended cell within subsecond times. Questions remain, however, about the origin of fluidity as a conserved parameter across timescales, the physical interpretation of its magnitude, and its potential use for high-throughput sorting and separation of interesting cells by mechanical means. Therefore, we exposed suspended CH27 lymphoma cells to various chemoenvironmental conditions—temperature, pharmacological agents, pH, and osmolarity—and measured cell fluidity with a non-contact technique to extend familiarity with suspended-cell mechanics in the context of both soft-matter physics and mechanical flow cytometry development. The actin-cytoskeleton-disassembling drug latrunculin exacted a large effect on mechanical behavior, amenable to dose-dependence analysis of coupled changes in fluidity and stiffness. Fluidity was minimally affected by pH changes from 6.5 to 8.5, but strongly modulated by osmotic challenge to the cell, where the range spanned halfway from solid to liquid behavior. Together, these results support the interpretation of fluidity as a reciprocal friction within the actin cytoskeleton, with implications both for cytoskeletal models and for expectations when separating interesting cell subpopulations by mechanical means in the suspended state.

 Please wait while we load your content...
Please wait while we load your content...