Micellization and related behavior of sodium dodecylsulfate in mixed binary solvent media of tetrahydrofuran (Tf) and formamide (Fa) with water: a detailed physicochemical investigation†
Abstract
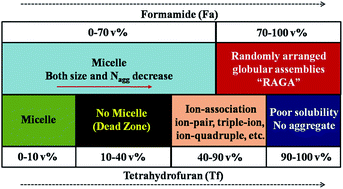
The detailed aggregation behavior of sodiumdodecyl sulfate (SDS) in tetrahydrofuran (Tf)–water (W) and formamide (Fa)–water (W) media at varied volume percent compositions has been investigated. Surface tension (ST), conductance (Cond), viscosity (Visc), isothermal titration calorimetry (ITC), nuclear magnetic resonance (NMR) and small angle neutron scattering (SANS) were used in this study. The presence of nonaqueous solvents affected the critical micelle concentration (CMC) of SDS, the counter-ion binding of the micelle, the energetics of the air/water interfacial adsorption and micellization of the amphiphiles in the bulk, the ion-association (ion-pair, triple-ion, quadruple, etc. between Na+ and DS− ions) as well as the weakly soluble (aggregation less) amphiphile solution. Tf has been observed to produce a “dead zone” or “non-micelle formation zone” in the mixed Tf–W domain of 10–40 vol%. Fa influenced the SDS aggregation up to 70 vol%, at higher proportions (below the Krafft temperature (KT)), instead of the micelle, “randomly arranged globular assembly” (RAGA) was formed. The correlation of the standard free energy of micellization (ΔG0m) with different solvent parameters (1) dielectric constant (ε), (2) viscosity (η0), (3) Reichardt parameter (ET(30)), (4) Gordon parameter (G), and (5) Hansen–Hildebrand hydrogen bonding parameter (δh) has been attempted. It has been found that δh produced a master correlation between ΔG0m and δh for different binary mixtures such as Tf–W, Fa–W, Ml–W and Dn–W.

 Please wait while we load your content...
Please wait while we load your content...