Using THz time-scale infrared spectroscopy to examine the role of collective, thermal fluctuations in the formation of myoglobin allosteric communication pathways and ligand specificity†
Abstract
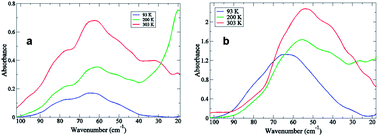
In this investigation we use THz time-scale spectroscopy to conduct an initial set of studies on myoglobin with the aim of providing further insight into the global, collective thermal fluctuations in the protein that have been hypothesized to play a prominent role in the dynamic formation of transient ligand channels as well as in shaping the molecular level basis for ligand discrimination. Using the two ligands O2 and CO, we have determined that the perturbation from the heme–ligand complex has a strong influence on the characteristics of the myoglobin collective dynamics that are excited upon binding. Further, the differences detected in the collective protein motions in Mb–O2 compared with those in Mb–CO appear to be intimately tied with the pathways of long-range allosteric communication in the protein, which ultimately determine the trajectories selected by the respective ligands on the path to and from the heme-binding cavity.

 Please wait while we load your content...
Please wait while we load your content...