Electrostatically driven lipid–lysozyme mixed fibers display a multilamellar structure without amyloid features†
Abstract
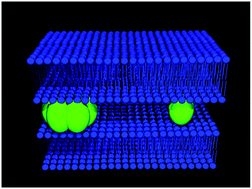
Understanding the interactions between anionic lipid membranes and amyloidogenic proteins/peptides is key to elucidate the molecular mechanisms underlying the membrane-driven amyloid fiber formation. Here, hen egg-white lysozyme was used as a model protein to test whether this same process also occurs with non-amyloidogenic lipid-binding proteins/peptides. A complementary set of biophysical techniques was employed to study the structure and dynamics of the lipid–lysozyme mixed fibers produced at a low lipid/protein molar ratio that have been proposed earlier to present “amyloid-like” characteristics. The multilamellar architecture of these elongated mesoscopic structures was established by performing time-resolved Förster resonance energy transfer measurements, at both bulk (ensemble) and single-fiber level. The predominantly oligomeric lysozyme and phospholipids were both found to display significantly decreased lateral mobility when embedded in these mixed fibers. Notably, two-photon microscopy of Laurdan revealed that a pronounced membrane surface dehydration/increased molecular interfacial packing was produced exclusively in these elongated mixed supramolecular fibers present in the highly polymorphic samples. Infrared spectroscopic studies of lysozyme in these samples further showed that this protein did not exhibit a rich β-sheet structure characteristic of amyloid fibrils. These results support the conclusion that negatively charged lipid membranes do not have the general ability to trigger amyloid fibril formation of non-amyloidogenic proteins.

 Please wait while we load your content...
Please wait while we load your content...