Observing the oxidation state turnover in heterogeneous iridium-based water oxidation catalysts†
Abstract
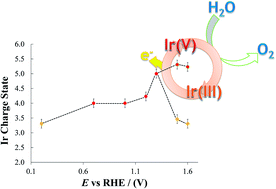
In this work the oxidation states assumed by Ir in oxide systems used as heterogeneous catalysts for water oxidation are determined by means of in situ X-ray Absorption Spectroscopy (XAS). Using a highly hydrated iridium oxide film allows the maximum number of Ir sites to be involved in the electrochemical processes occurring at the catalysts during water oxidation (oxygen evolution reaction, OER). X-ray Absorption Near Edge Structure (XANES) spectra clearly indicate the co-existence of Ir(III) and Ir(V) at the electrode potentials where OER occurs. This represents a fundamental step both in the understanding of the water oxidation mechanism catalysed by heterogeneous Ir oxide systems, and in the possible tailoring of electrocatalysts for OER.

 Please wait while we load your content...
Please wait while we load your content...