Addition of pnictogen atoms to chromium(ii): synthesis, structure and magnetic properties of a chromium(iv) phosphide and a chromium(iii) arsenide†‡
Abstract
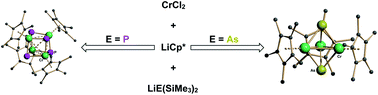
Chromium(II) chloride reacts with lithium pentamethylcyclopentadienide (LiCp*, Cp* = C5Me5) and LiE(SiMe3)2 (E = P or As) to give the heterocubane chromium phosphide [(η5-Cp*Cr)(μ3-P)]4 (1) or the chromium arsenide cage [(η5-Cp*Cr)3(μ3-As)2] (2), respectively. The crystal and molecular structures of both compounds have been determined by X-ray crystallography. The reactions represent unusual oxidative additions of pnictogen atoms to chromium(II), which in the case of 1 results in the formation of the unusual chromium(IV) formal oxidation state, and in the case of 2 provides access to chromium(III). Compound 1 is also a rare example of a transition metal μ3-phosphide-ligated cubane. Magnetic susceptibility and magnetization measurements, with support from DFT calculations, indicate S = 2 and S = 9/2 ground states for 1 and 2, respectively, which can be rationalized by considering the electronic structure in terms of chromium–chromium bonding.

 Please wait while we load your content...
Please wait while we load your content...