Photoinduced electron transfer in a carbon nanohorn–C60 conjugate†
Abstract
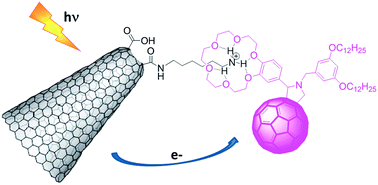
A nanohybrid combining two allotropic forms of carbon, namely carbon nanohorns (CNH) and C60, has been obtained from a C60 derivative bearing a benzocrown ether subunit (crown–C60) and a CNH functionalized with NH3+ groups (CNH-sp-NH3+F−) through ammonium–crown ether interactions. The resulting CNH–C60 nanohybrid has been characterized by Raman and XPS spectroscopies, thermogravimetric analysis (TGA) and high-resolution transmission electron microscopy (HR-TEM). The photophysical properties of the CNH–C60 conjugate have been investigated in benzonitrile. Femtosecond laser flash photolysis measurements revealed the occurrence of an efficient electron transfer from the singlet excited state of the C60 moiety to the CNH with a rate constant of 6.5 × 1010 s−1 to produce a radical ion pair, which decayed by charge recombination with a lifetime of 1.0 ns to afford the triplet excited state of CNH-sp-NH3+F− and crown–C60. The two carbon nanoforms play therefore complementary roles in the CNH–C60 conjugate, the CNH acting as an electron donor and C60 as an electron acceptor.

 Please wait while we load your content...
Please wait while we load your content...