β-Lactonization of fluorinated porphyrin enhances LDL binding affinity, cellular uptake with selective intracellular localization†
Abstract
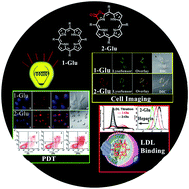
Porpholactones, with one porphyrin β,β′-double bond replaced by a lactone moiety, possess unusual electronic states and photophysical properties between porphyrin and chlorin. However, such optical properties and unique structural characteristics have rarely been investigated within biological studies. In this work, we report that β-lactonization of porphyrin decreases the lipophilicity (log Po/w), due to the lower pKa value of pyrrollic nitrogen arising from the electron-withdrawing lactone moiety, and increases singlet oxygen quantum yields. Attaching glucose conjugates to the fluorinated porpholactones results in a high binding affinity with low density lipoprotein (LDL), which facilitates cellular-uptake efficacy through an LDL-dependent pathway and selective intracellular localization (lysosome). More importantly, β-lactonization of porphyrin endows higher photocytotoxicity against Hela cells through apoptosis, this is highly associated with increasing intracellular ROS levels. These results increase the understanding of the relationship between the basic structural characteristics and biological activity of porphyrins containing a non-pyrrolic moiety and will be important to expand the scope of β-modified porphyrinoids as potential photosensitizers for photodynamic therapy.

 Please wait while we load your content...
Please wait while we load your content...