Coordinating ligand functionalized AgNPs for colorimetric sensing: effect of subtle structural and conformational change of ligand on the selectivity†
Abstract
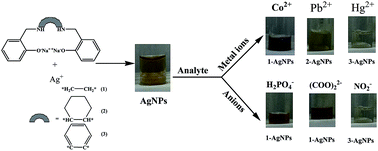
Metal coordinating C2-symmetric phenolic chelating ligands (1–3) were used to prepare surface functionalized AgNPs for selective colorimetric sensing of metal cations and anions. The effect of subtle structural and conformational change of 1–3 on the colorimetric sensing was also explored. UV-Visible, IR spectroscopy and HR-TEM techniques were used to characterize AgNPs. Interestingly 1-, 2- and 3-AgNPs showed different colorimetric sensing of metal cations and anions. Aliphatic flexible diamine based 1-AgNPs showed selective sensing of Co2+ ions (10−7 M), oxalate and dihydrogen phosphate anions (10−6 M). Co2+ to 1-AgNPs produced a brownish-orange colour whereas oxalate and phosphate resulted in a brownish-pink colour due to aggregation. Alicyclic diamine based 2-AgNPs exhibited selective precipitate formation for Pb2+ ions without showing any anion sensing. The rigid diamine based 3-AgNPs showed selective decolourisation of the yellow colour for Hg2+ and nitrite anions due to amalgamation and oxidation. Thus phenolic chelating ligands that have been used to generate versatile coordination networks with metal ions offer a simple way to develop AgNPs based potential colorimetric sensors for environmental screening.

 Please wait while we load your content...
Please wait while we load your content...