Theoretical calculation based synthesis of a poly(p-phenylenediamine)–Fe3O4 composite: a magnetically recyclable photocatalyst with high selectivity for acid dyes†
Abstract
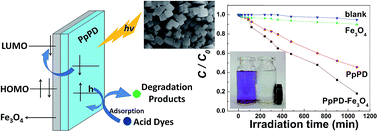
A magnetically recyclable and highly photocatalytic poly(p-phenylenediamine)–Fe3O4 (PpPD–Fe3O4) composite photocatalyst was synthesised by a one-step chemical oxidation polymerisation based on theoretical calculation results. The band gap and selectivity of PpPD for acid dyes were studied by theoretical calculations. The calculation results showed that the PpPD molecules exhibited better conjugacy than PANI and that the band gap was more fit for use as a photocatalyst. The photocatalytic activities were evaluated by the degradation of various dyes. The stability and high selectivity of this composite was remarkable. The combination of Fe3O4 with PpPD lead to high photocatalytic activity in the degradation of the dyes under both ultraviolet, and visible light, irradiation, although Fe3O4 alone was inactive as a visible-light-driven photocatalyst.


 Please wait while we load your content...
Please wait while we load your content...