Influence of the degree of polymerisation and of the architecture on the elastic properties of new polyurea elastomers†
Abstract
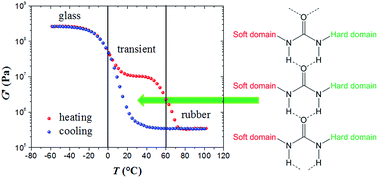
The elastic properties of new polyurea elastomers have been studied by varying the segmental molecular weight and the chemical nature of the polymer end groups. Three different types of elastomers were synthesized leading to three different types of response. The elastomers with a high degree of polymerisation and primary amines as terminal groups show two plateaus: at high temperature, the common permanent plateau related to the rubber behaviour of elastomeric systems and, at low temperature, a transient plateau associated with the hydrogen bonding of the urea motives occurring in the interfacial zone between the soft polyetheramine and the hard crosslinker domains. The elastomers with a low degree of polymerization and primary amines as terminal groups show that the transient plateau is masked by the glassy plateau because the hydrogen bonds occur in the same temperature range as the glass transition effects, except for very slow heating rates for which the transient network can be resolved. Lastly, the elastomers with no hydrogen bonding just show the common step in the elastic behaviour from the rubbery to the glassy state.

 Please wait while we load your content...
Please wait while we load your content...