Switchable synthesis of 2,5-dimethylfuran and 2,5-dihydroxymethyltetrahydrofuran from 5-hydroxymethylfurfural over Raney Ni catalyst†
Abstract
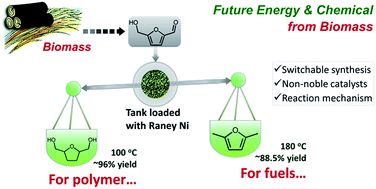
Raney-type metals (Cu, Co and Ni) were employed to catalyze hydrogenation of 5-hydroxymethylfurfural. Switchable synthesis of 2,5-dimethylfuran and 2,5-dihydroxymethyltetrahydrofuran was achieved with 96% and 88.5% yield respectively over Raney Ni, demonstrating high feasibility for industrialization. The excellent yields can be explained by the fact that Raney Ni facilitates the hydrogenation reaction but has limited deoxygenation ability at low temperature, while high temperature promotes the deoxygenation step. The reaction pathway was analyzed by time course experiments and HMF hydrogenation over model catalysts was performed. The reaction mechanism related to the respective catalytic sites was discussed and proposed, which has great implications in the design of efficient and non-noble metal catalysts.

 Please wait while we load your content...
Please wait while we load your content...