Use of amine electride chemistry to prepare molybdenum disulfide intercalation compounds
Abstract
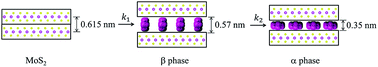
Electride solutions, formed by dissolution of Na(m) in ethylenediamine (en), are used to generate MoS2 intercalation compounds. Two new intercalation compounds were obtained, labeled α and β phases, containing both Na and en as intercalate and cointercalate respectively. The intercalation reaction proceeds via formation of a kinetic product, the β phase, and subsequent generation of a stable thermodynamic product, the α phase. The β and α phases have interlayer distances, dIC, of 1.19 and 0.97 nm, respectively, which are ascribed to a perpendicular or parallel orientation of en within the intercalate galleries. Product compositions, morphologies and spectrochemical features are obtained from thermogravimetry, SEM and Raman measurement. Electrochemical reduction of MoS2 in an en-based electrolyte is investigated, but does not show the same intercalation reactions. The chemistry observed is compared with that previously reported for this and other MS2 hosts, and with graphite intercalation.
- This article is part of the themed collection: Materials for Energy storage

 Please wait while we load your content...
Please wait while we load your content...