Study on the relation between pore size and supercapacitance in mesoporous carbon electrodes with silica-supported carbon nanomembranes†
Abstract
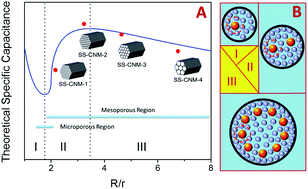
Electrochemical capacitors (ECs) have traditionally been considered as standing at the opposite end against batteries in energy–power diagram. They charge and discharge faster than batteries but are limited by much lower energy density. By optimizing the pore structure of porous electrode materials, the performance of ECs could overcome this limitation. However to date, no study has addressed the complex relationship between the texture parameters of the electrode materials and the supercapacitance of ECs. Using silica-supported carbon nanomembranes, four electrode materials with similar pore geometry are generated. The electrodes with a pore size of 4.14 nm shows the highest capacitance of 305 F g−1 in aqueous electrolytes. A new model is developed to simulate the accommodation of the solvated ions at the electrode surface. The simulation reveals that the optimal capacitance of ECs can be achieved using porous carbon electrode materials with open pores of 3.0–5.0 nm.

 Please wait while we load your content...
Please wait while we load your content...