Synthesis of purely silica MFI-type nanosheets for molecular decontamination
Abstract
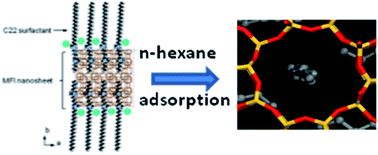
Conventional syntheses of zeolites generally lead to the formation of crystals whose sizes are of the order of several microns which is not detrimental in a large number of industrial applications. However, the capacity and kinetics of pollutant adsorption which are sensitive to diffusion phenomena, surface and porous volume could potentially be improved by the use of nanocrystal or hierarchical zeolites (micro/mesoporous or micro/macroporous). Indeed, zeosil nanosheets hold great potential because of their small size and their high porous volume that promote access of pollutants and increase the adsorption capacity. Herein, silicalite-1 zeosil with a lamellar morphology was successfully synthesized under hydrothermal conditions (110 °C, 10 days) using diquaternary ammonium as structuring agent. Compared to the conventional silicalite-1 material, the porous volume of the synthesized nanosheets determined from the N2 adsorption–desorption isotherms was found to be multiplied by 3.5 (0.62 cm3 g−1) without altering the microporous volume (0.18 cm3 g−1). This result was also confirmed by the increase of the n-hexane adsorption capacity and kinetics in the silicalite-1 nanosheets compared to the conventional silicalite-1. This approach indicates a new way for obtaining zeosil materials of controlled sizes and shapes for molecular decontamination.
- This article is part of the themed collection: Porous Materials (FEZA 2014)

 Please wait while we load your content...
Please wait while we load your content...