First-principles study of crystalline CoWO4 as oxygen evolution reaction catalyst
Abstract
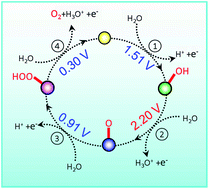
Cobalt tungstate (CoWO4) was recently reported as a catalyst that drives the oxygen evolution reaction. Interestingly, the catalytic activity of crystalline CoWO4 is orders of magnitude lower than its amorphous counterpart. In order to better understand the mechanical origin of these observations, herein we study the oxygen evolution on the crystalline CoWO4 surface using first-principles calculations. By comparing the calculated data with experimental measurements, we demonstrate that GGA + U calculations with U = 4.5 effectively predict the structural and electronic properties of crystalline CoWO4. With this U value the overpotential for oxygen evolution on the crystalline CoWO4 surface is 0.97 V. This prediction agrees fairly well with experimental estimation, and is consistent with the low catalytic activity of crystalline CoWO4. By a comprehensive analysis of the reaction process, such high overpotential is attributed to the instability of oxygen radicals formed during the oxygen evolution. Our results highlight the importance to stabilize oxygen radicals during oxygen evolution reaction. It also paves the road for future optimization of CoWO4 as oxygen evolution catalyst.

 Please wait while we load your content...
Please wait while we load your content...