Efficient Ru–Fe catalyzed selective hydrogenolysis of carboxylic acids to alcoholic chemicals†
Abstract
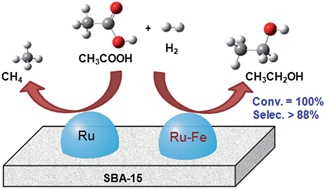
Supported bimetallic Ru–Fe catalysts were prepared using a step-deposition–reduction method. The selective hydrogenolysis of acetic acid to ethanol was investigated as a reaction, which is considered to be related to the transformation of biomass-derived carboxylic acids to fuels and value-added chemicals. An SBA-15-supported Ru–Fe catalyst displayed significant improvements in catalytic performance for the hydrogenolysis of acetic acid to ethanol compared with monometallic catalysts and that with SiO2 as a carrier. When the Ru/Fe atomic ratio was set at 2/1, the prepared catalyst could give a nearly 100% conversion of acetic acid and 88% selectivity to ethanol. The catalyst showed considerable stability in terms of structure and performance for a long-term run on stream. Characterization results indicated that a small portion of Fe species was alloyed with Ru, whereas the other portion of Fe species, likely FeO1+x (0 < x < 0.5), was dispersed on the catalyst surfaces. The Fe species were crucial for the stabilization of Ru–Fe bimetallic nanoparticles and activation of acetic acid molecules in the hydrogenolysis reaction. Moreover, several other carboxylic acids, such as propionic acid, levulinic acid, and lactic acid, could also be efficiently converted to their corresponding alcoholic chemicals or lactone using the optimized Ru–Fe/SBA-15 catalyst under relatively mild conditions.

 Please wait while we load your content...
Please wait while we load your content...