Synthesis and properties of a meso- tris–ferrocene appended zinc(ii) porphyrin and a critical evaluation of its dye sensitised solar cell (DSSC) performance†
Abstract
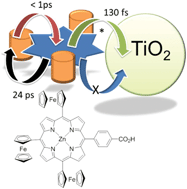
A zinc(II) porphyrin derivative, F3P, was prepared containing a single ferrocene group appended at three of the meso positions. The final meso position contains a benzoic acid unit, and was designed to be an anchoring moiety to a semiconductor surface. The cyclic voltammogram for the porphyrin derivative reveals that the three ferrocene units are oxidised and reduced at E1/2 = +0.04 V vs. Fc+/Fc. Other redox responses are porphyrin-based at +1.1 V vs. Fc+/Fc (irreversible) and −1.87 V and −2.20 V vs. Fc+/Fc (both quasi-reversible). The room temperature Mössbauer spectrum for F3P comprises a doublet (δ Fe 0.44 mm s−1; ΔEQ 2.35 mm s−1; peak width 0.27 mm s−1). The data are consistent with the ferrocene groups non-interacting with the porphyrin moiety in the ground state. Excitation of the compound in THF with an ultra-short laser pulse produces in less than 1 ps the charge separated state which comprises Fc+˙–porp−˙ and decays back to the ground state in 24 ps. Roughly the same behaviour is observed for the dye absorbed on TiO2, the only slight difference is the appearance of a long-lived component in the decay records. The electron for the porphyrin radical anion does not inject into the conduction band of the TiO2, only the porphyrin excited state participates in any electron injection process. Partly because of this factor the performance of the dye attached to TiO2 in a DSSC device is rather limited (JSC = 0.068 mA cm−2, VOC = 283 mV, FF = 0.42, η = 0.008%).

 Please wait while we load your content...
Please wait while we load your content...