A green process for the production of butanol from butyraldehyde using alcohol dehydrogenase: process details†
Abstract
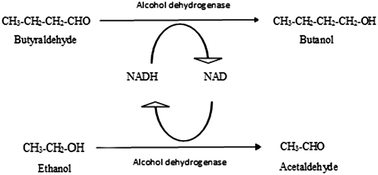
Depletion of energy sources has drawn attention towards production of bio-butanol by fermentation. However, the process is constrained by product inhibition which results in low product yield. Hence, a new strategy wherein butanol was produced from butyraldehyde using alcohol dehydrogenase and NADH as a cofactor was developed. Butyraldehyde can be synthesized chemically or through fermentation. The problem of cofactor regeneration during the reaction for butanol production was solved using substrate coupled and enzyme coupled reactions. The conventional reaction produced 35% of butanol without regeneration of cofactor using 300 μM NADH. The process of substrate coupled reaction was optimized to get maximum conversion. NADH (30 μM) and 100 μg per ml of alcohol dehydrogenase (320 U mg−1) could convert 17.39 mM of butyraldehyde to butanol using ethanol (ratio of butyraldehye to ethanol 1 : 4) giving a maximum conversion of 75%. The enzyme coupled reaction under the same conditions showed only 24% conversion of butyraldehyde to butanol using the glutamate dehydrogenase-L-glutamate enzyme system for the regeneration of cofactor. Hence, substrate coupled reaction is suggested as a better method over the enzyme coupled reaction for the cost effective production of butanol.

 Please wait while we load your content...
Please wait while we load your content...