Chiral transmission to crystal photodimerizations of leucine–methionine quasiracemic assemblies†
Abstract
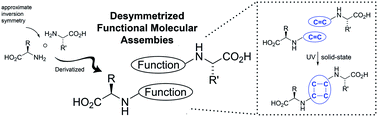
We report the deliberate use of molecular shape as a structural tool for generating homochiral photoreactive crystalline materials. Building on the crystal packing tendencies of amino acid quasiracemates to form near inversion related motifs, L-leucine and D-methionine were decorated with sulfonamidecinnamic acid groups. When co-crystallized, the ‘fish hook’ shaped components organize into supramolecular dimers with favorable olefin⋯olefin spacing (3.71 and 4.09 Å). UV exposed samples were processed as single-crystal-to-single-crystal transformations and showed quantitative conversion to the expected enantiopure photodimerization product. Similar crystal motifs and reaction outcomes were achieved using the racemic counterparts suggesting the importance of best-fit scenarios directed by the complementary features of molecular shape and non-bonded contacts.

 Please wait while we load your content...
Please wait while we load your content...