Fast hydrogen release under moderate conditions from NaBH4 destabilized by fluorographite†
Abstract
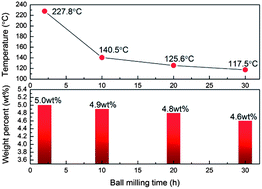
A simple approach to dramatically enhance the dehydrogenation properties of sodium borohydride is achieved by ball milling NaBH4 with fluorographite (FGi). It was found that the ball-milled NaBH4–FGi composite starts to release hydrogen without impurity gas at a lower temperature of 125 °C, and obtains a hydrogen desorption capacity of ca. 4.8 wt% below 130 °C in seconds, which is improved markedly compared to the ball-milled pristine NaBH4. The significant thermodynamic and kinetic improvement of the NaBH4–FGi composite can be ascribed to the reaction between NaBH4 and FGi as well as the formation of micro-scale NaBH4. Moreover, since the dehydrogenation process of NaBH4–FGi composite is exothermal, the fully reverse reaction is not feasible. In-depth investigations show that the partial rehydrogenation is due to the formation of Na2B12H12 and another new borohydride.

 Please wait while we load your content...
Please wait while we load your content...